Biotle Announces Preclinical PK/PD Results for FcRn Inhibitor BTL-203; Modeling Supports Once-Every-Four-Weeks Subcutaneous Dosing in Humans
HANGZHOU, China — September 8, 2025 — Biotle today announced positive preclinical pharmacokinetic/pharmacodynamic (PK/PD) results for BTL-203, the company’s half-life–extended neonatal Fc receptor (FcRn) inhibitor. In cynomolgus monkeys, BTL-203 produced deep and sustained reductions in total IgG after single-dose intravenous (IV) or subcutaneous (SC) administration. Using a semi-mechanistic FcRn–IgG turnover model calibrated to these data, simulations project that subcutaneous dosing every four weeks (Q4W) could maintain clinically meaningful IgG suppression in humans.
“These encouraging data, together with our translational modeling, suggest BTL-203 could offer once‑every‑four‑weeks subcutaneous dosing—meaningfully less frequent than today’s approved FcRn regimens, which are administered in weekly cycles. We believe this convenience, alongside potent IgG lowering, positions BTL-203 as a potential best‑in‑class therapy, subject to confirmation in clinical studies.”
— Dr. Jiansong Yang, CEO of Biotle
Nonclinical Highlights
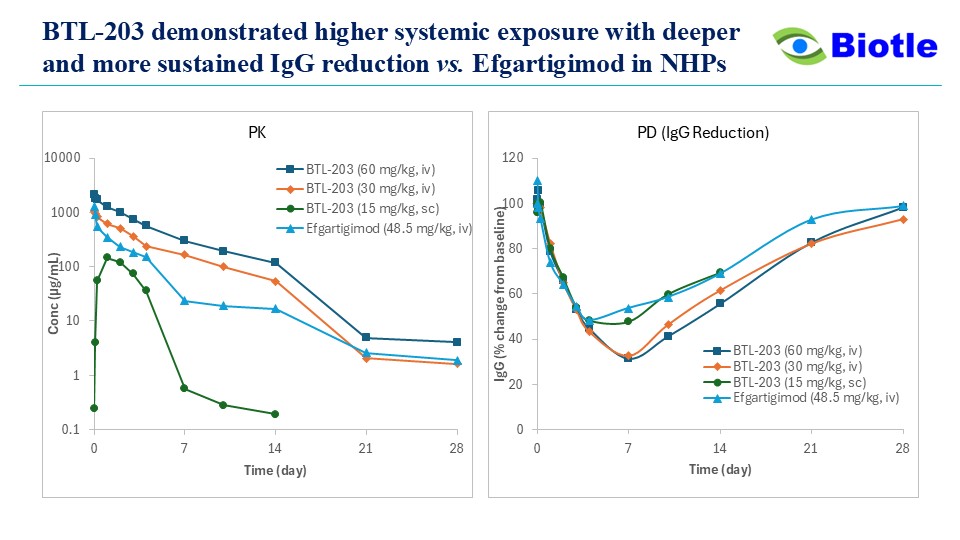
- Robust IgG lowering vs comparator: Single IV doses of BTL-203 (60 or 30 mg/kg) achieved higher systemic exposure and deeper, more sustained total‑IgG reductions than efgartigimod IV (48.5 mg/kg) in the study; nadir decreases were ~67–69% with recovery by ~4 weeks.
- SC administration: BTL-203 15 mg/kg SC produced total‑IgG reduction comparable to efgartigimod 48.5 mg/kg IV.
- Translational projection: Population PK/PD modeling suggests Q4W SC dosing in humans could sustain IgG lowering within a target therapeutic window; this will be tested prospectively in clinical trials.
- Safety & tolerability: BTL-203 was well-tolerated in cynomolgus monkeys. At 15 mg/kg SC, albumin decreases and LDL‑C changes were minimal and reversible.
Potential differentiation. Approved FcRn therapies today are delivered in weekly (SC) or biweekly (IV) treatment cycles. If confirmed clinically, a Q4W SC maintenance regimen would represent a less frequent dosing schedule for an FcRn inhibitor.
About BTL-203
BTL-203 is an FcRn inhibitor engineered for extended exposure and infrequent subcutaneous dosing. By blocking FcRn, BTL-203 accelerates IgG catabolism—a validated mechanism across multiple IgG‑mediated diseases. Biotle is advancing BTL-203 toward clinical development (planned IND submission in Q2 2026) and is engaging potential partners to accelerate global development and commercialization.
Business Development: contact us to explore partnering opportunities.

