Our Pipeline
Biotle is advancing a robust pipeline of innovative antibody-based therapeutics designed to deliver best-in-class and first-in-class treatments for cancer patients worldwide. Our programs span multiple stages of development, targeting various tumor types and leveraging our proprietary TriME® technology platform.
We are committed to translating our scientific breakthroughs into transformative therapies that address significant unmet medical needs.
Pipeline Overview
The following chart provides an overview of our current clinical and preclinical programs. Strategy Validated targets × Differentiated design = higher probability of technical and commercial success.
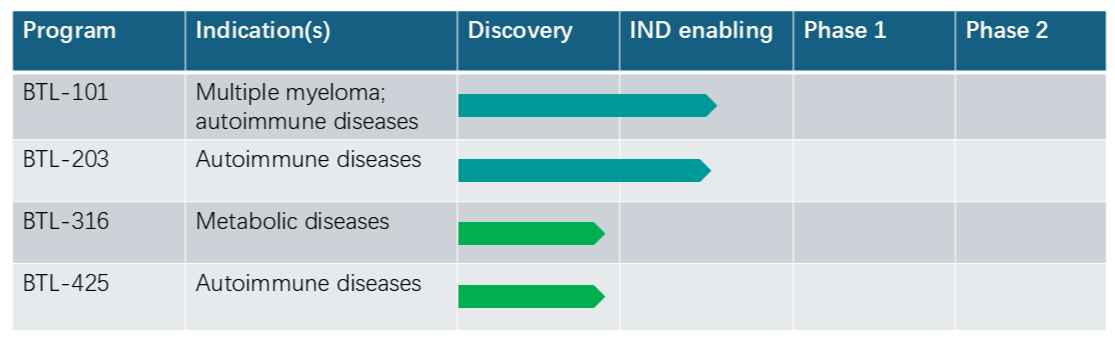
BTL-101 (targets undisclosed)
BTL-101 is our pioneering trispecific T-cell engager that simultaneously targets CD3ε and two tumor-associated antigens (TAAs) to address tumor heterogeneity in relapsed or refractory multiple myeloma and select autoimmune indications. Built on Biotle’s proprietary TriME® platform, BTL-101 is positioned as a potential best‑in‑class therapy:
- Simultaneous targeting of CD3ε and two tumor-associated antigens (TAAs) to overcome antigen escape and resistance.
- Fully human, high-affinity binding domains with low CD3ε affinity to reduce cytokine-release risk.
- Fc LALAPG silencing to prevent unwanted T-cell clearance.
- High subcutaneous bioavailability (~100% in mice) and stable high-concentration formulation (>100 mg/mL).
- Projected once-every-4-weeks subcutaneous dosing; strong CMC profile (7–8 g/L titers; 4-week stability at 40 °C).
- Demonstrated potent, precise tumor-cell killing with minimal off-target effects in preclinical models.
BTL-203 (a potential best-in-class FcRn inhibitor)
BTL-203 is our next-generation bispecific antibody designed for deep, durable IgG reduction with convenient once-every-4-weeks subcutaneous dosing for autoimmune diseases. Advantages include:
- Extended exposure and stabilized effect.
- Deep & durable effect linked to superior outcomes in autoimmune diseases.
- Improved convenience and compliance vs. weekly or biweekly dosing by competitors.
- Supports broad indication expansion in chronic autoimmune diseases.
Partnering
We are open for partnering opportunities.
